top of page
01. Nasal COVID vaccine available both as a primary dose and a heterologous booster.
December 24, 2022 at 4:21:14 AM
Vaccination
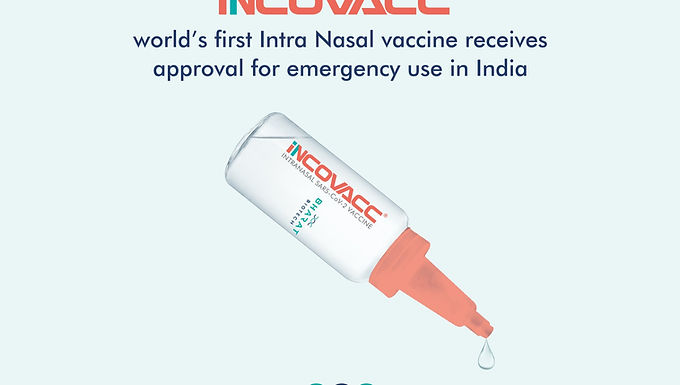
iNCOVACC, co-developed by Hyderabad-based Bharat Biotech and the U.S.-based Washington University, was approved in November for ‘restricted emergency use’ for those aged 18 or older.
Approved as a primary dose after Phase-3 trials, and was separately tested for safety and immunogenicity as a booster. Unlike Covaxin, which is an inactivated coronavirus injected along with an adjuvant, iNCOVACC is an adenovirus-vectored vaccine with a “prefusion stabilised spike protein”, which is more like the technology employed in the Covishield vaccines.The nasal vaccine would be available via the CoWIN website.
bottom of page
.png)